Answers to Dr. Wimley's Homework problems
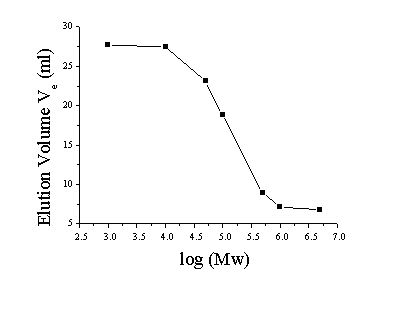
You have purified an active enzyme. (It gives a single peak using several non-denaturing liquid chromatographic methods). You run a set of molecules of known molecular weight along with your protein through a gel filtration column and record the following elution volumes.
Mw
Volume (ml)
5,000,000 6.8
1,000,000 7.1
500,000 8.9
100,000 18.8
50,000 23.1
10,000 27.5
1,000
27.7
Your protein elutes at 13.4 ml
A) What is the approximate molecular weight of your protein?
B) What is the columns void volume? Its included volume?
C) What range of molecular weight can be separated on this column?
Answers: The anwers to all three parts of
this question can be obtained by plotting the data like I plotted it on
my handouts:
First start with elution volume on the Y-axis
and log(MW) on the X-axis. You will notice that there are two plateaus.
The one at 6.8 ml is the Void Volume (Vo) and the one at 27.7
ml is the Total Volume (Vt).

You should have gotten the following:
Mw
log(Mw)
Volume (ml) Sigma
5,000,000
6.699
6.8
0.000
1,000,000
6.000
7.1
0.014
500,000
5.699
8.9
0.100
100,000
5.000
18.8
0.574
50,000
4.699
23.1
0.780
10,000
4.000
27.5
0.990
1,000
3.000
27.7
1.000
Unknown protein
13.4
0.316
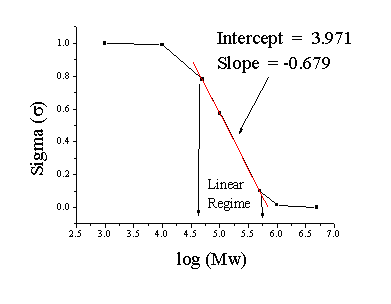
Now plot log(Mw) versus sigma (shown below). Three of the points fall on a straight line. The Log(Mw) of your protein can be calculated from the slope and intercept of that line or by interpolating between points. The unknown sigma of 0.316 corresponds to a log(Mw) of 5.38 or Mw ~ 240,000. The linear regime falls between log(Mw) = ~4.6 and ~5.7, which corresponds to Mw = 40,000 - 500,000. This is the the molecuar weight range that can be separated on this column.
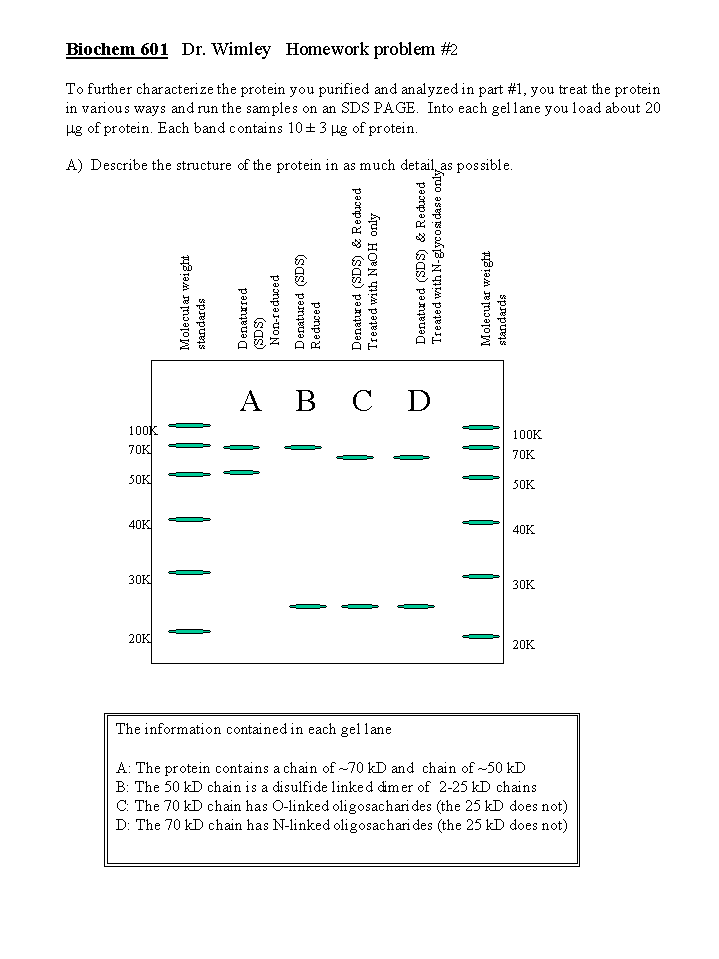
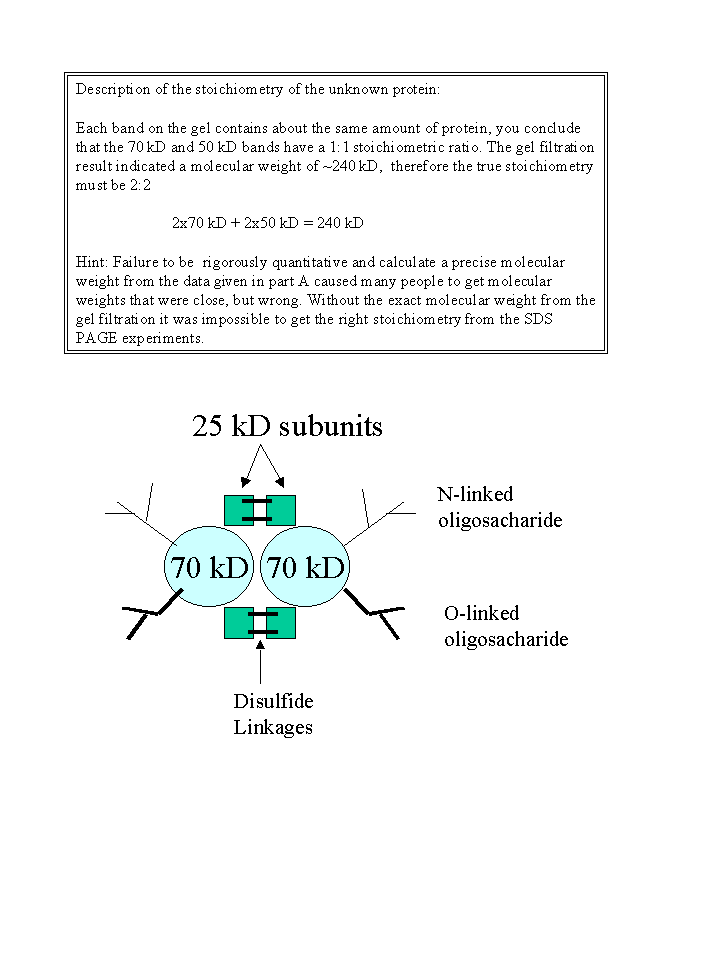
Answers to Dr. Wimley's Homework #2.
Send Comments, Questions, Corrections or Complaints to Dr. Wimley