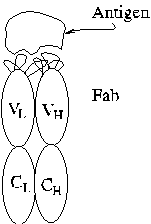Immunoglobulin Structure
Last modified 10/11/00.
Dr. Landry
Rm. 6055
landry@tulane.edu
Objectives
-
Become familiar with some immunology terminology.
-
Describe the domain structure of a molecule of immunoglobulin G (IgG).
-
Appreciate the structural details of antibody specificity and diversity.
-
Summarize structural and functional distinctions of the different antibody
classes.
-
Realize that members of the immunoglobulin superfamily of proteins have
diverse functions yet share the immunoglobulin fold.
Additional Resources
Glossary of Terms
-
antibody (immunoglobulin, Ig)
-
a protein that associates non-covalently with a foreign substance and initiates
a process that eliminates the substance from the organism
-
epitope
-
antibody-binding site on molecule (not necessarily the antigen) that reacts
with antibody
-
antigen
-
material that elicits antibody production
-
hapten
-
a small molecule that alone cannot elicit antibody production, but when
attached to a larger molecule can act as an antigenic determinant
and elicit antibody synthesis
-
Ig heavy chain (H)
-
larger of the two immunoglobulin chain types, approximately 50kDa in IgG,
isotype (gamma, alpha, delta, epsilon or mu) determines immunoglobulin
class (G, A, D, E or M, respectively)
-
Ig light chain (L)
-
smaller of the two immunoglobulin chain types, approximately 25kDa, either
of two isotypes (kappa or lambda) found in all antibody classes
-
Fab fragment
-
antigen-binding fragment of Ig generated by proteolysis with papain
-
Fc fragment
-
"crystallizable" fragment of Ig generated by proteolysis with papain, mediates
phagocytosis, triggers inflammation, targets Ig to particular tissues
-
complementarity determining regions (CDRs)
-
hypervariable loops in the immunoglobulin polypeptides that form the antigen
binding site, 3 regions of 5-7 residues in L and 3 or 4 regions of 6-17
residues in H.
Immunoglobulin Structure
Many features of Ig structure and Antigen-antibody interaction can be examined
in a kinemage.
Antibody molecules of type IgG are composed of four polypeptide chains,
two identical copies of each a light chain (L) and heavy chain (H). The
four chains are covalently bonded together by disulfide bonds. A small
amount of N-linked carbohydrate (CHO) is attached to H. Hypervariable complementarity
determining regions that determine the specificity of the antibody
lie in the amino-terminal domains of L and H.
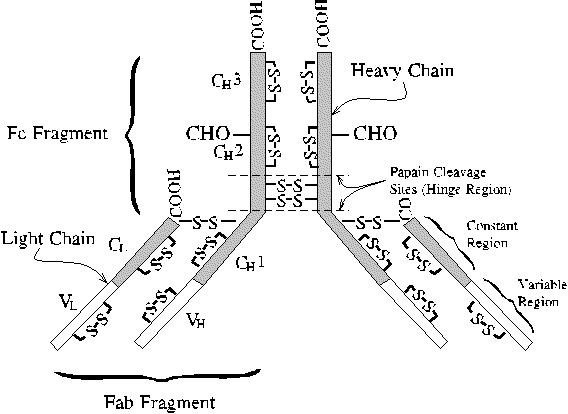
Fab and Fc fragments of IgG
Antibody functions can be separated into two proteolytic fragments by cleavage
with the enzyme papain in the hinge region. The
Fab fragment
retains the antigen-binding activity, binding to a monovalent antigen with
an affinity nearly as high as that of the entire antibody. The Fc fragment
mediates class-specific functions, such as complement fixation,
exhibited by IgG and IgM antibodies, in which a cascade of proteolytic
activation steps (analogous to the clotting cascade) leads to lysis of
the intruding cell.
The Immunoglobulin Fold
Despite their disparate functions, both Fab and Fc fragments contain domains
of similar three-dimensional structure. The shared domain defines the immunoglobulin
fold. The Ig fold is composed of all beta sheet secondary structure
and contains one disulfide bond. L has two immunoglobulin domains, one
variable (V) and one constant (C); whereas H has four domains, one variable
and three constant. The variable domains are so named because antibodies
of different antigen specificity have different sequences in the complementarity
determining regions (CDRs) of the variable domains. The CDRs are located
in loops connecting beta strands of the variable Ig domains. When L and
H pair up in the antibody structure, the CDR-containing loops are brought
together to form the antigen binding site.
Use the following link to view a ribbon
diagram of an Fab fragment co-crystallized with its hapten. [This is
an example of a catalytic antibody that was designed to have enzymatic
activity. The hapten is a transition-state analog that mimics the theoretical
transition state structure for enyzmatic hydrolysis of an ester linkage.]
Use the following link to view a ribbon
diagram of one Fc chain co-crystallized with a fragment of an IgG-binding
bacterial protein (Staphylococcus aureus protein A). Note that
this fragment contains two constant immunoglobulin domains. The ability
of Protein A to bind to IgG has been exploited for many years to detect
the presence of antigen-antibody complexes, such as in enzyme-linked immunosorbent
assays (ELISA) or Western blots. However, it is now more common to use
anti-IgG produced by an heterologous organism, e.g. goat anti-rabbit
IgG.
Antigen-antibody Interaction
precipitin reaction
Antibodies are divalent with respect to antigen binding and most antigens
are multivalent (for example, numerous antigen molecules on a foreign cell
surface); thus, a specific antigen-antibody reaction yields a crosslinked
lattice that forms a precipitate. Flexibility in the hinge region of antibodies
may afford a greater ability to make such multivalent contacts with different
antigen materials.
features of the interface
The same types of interactions stabilize Ag-Ab binding as stabilize the
folded structure of proteins, i.e., hydrophobic interactions, hydrogen
bonds, van der Waals contacts, and electrostatic interactions. The two
surfaces generally have complementary shapes with respect to the placement
of grooves and bumps, maximizing affinity and specificity.
Immunoglobulin Class Distinctions
-
IgG
-
H type gamma; most abundant Ig; primarily found in plasma and interstitial
fluid; transported across placenta; synthesis during immune response peaks
4-5 days after peak of IgM
-
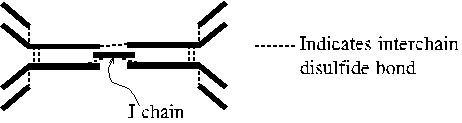
IgA
-
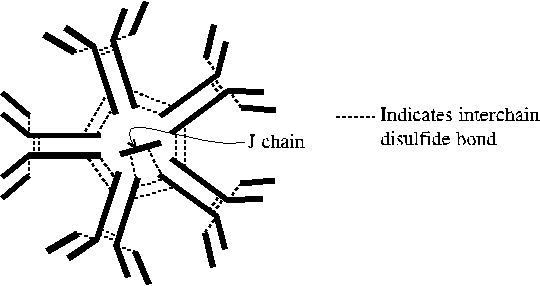
H type alpha; with J chain forms dimer or trimer of the basic immunoglobulin
structure; primarily found in mucosal secretions (intestine, sweat, saliva,
tears, milk); blocks pathogen passage across endothelium
-
IgM
-
H type mu; primarily found in plasma; first to appear in immune
response
-
IgE
-
H type epsilon; low abundance; important in allergy; targeted at
parasites?
-
IgD
-
H type delta; low abundance; function unknown
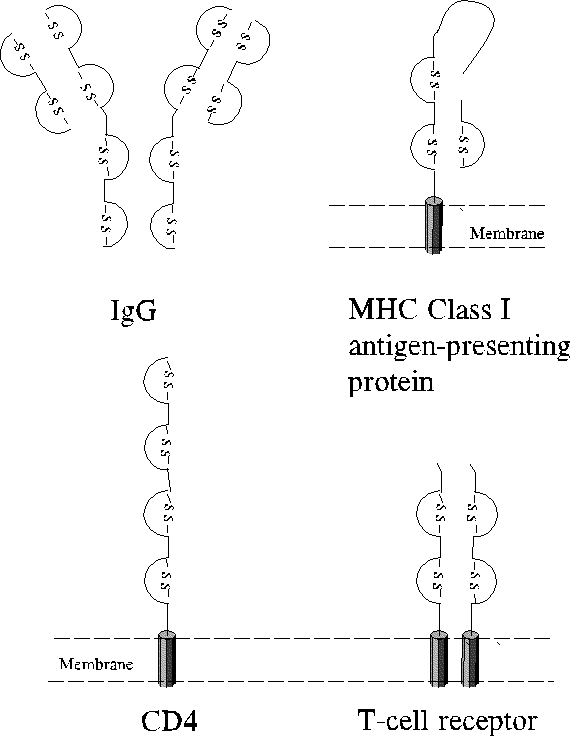
The Immumoglobulin Superfamily of Proteins
Immunoglobulin domains occur in diverse proteins involved in the immune
response. Some examples are illustrated below.
End of Document